Expression and clinical significance of adaptor-related protein complex 1 subunit Mu2 in colorectal carcinoma
-
摘要:目的
探讨适配器相关蛋白复合物1亚基Mu2(AP1M2)表达与结肠癌患者临床病理特征的相关性。
方法收集2017年1—12月连云港市第二人民医院行根治性手术治疗的97例结肠恶性肿瘤患者的癌组织及癌旁组织。收集患者临床病理特征资料,采用免疫组化法检测结肠癌组织与癌旁组织中AP1M2蛋白的表达水平,采用酶联免疫吸附测定法检测AP1M2在血清中表达情况。分析AP1M2蛋白表达与结肠恶性肿瘤患者临床病理特征的关系; 采用Kaplan-Meier法分析结肠癌患者术后5年生存情况的影响因素; 采用生物信息学分析AP1M2涉及的蛋白及相关通路情况。
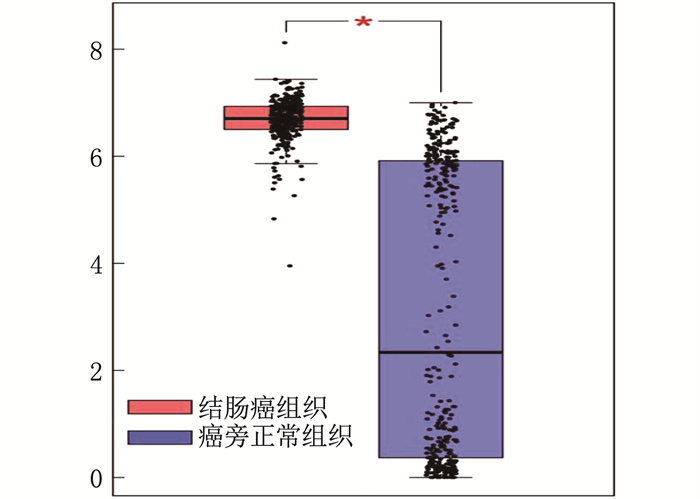
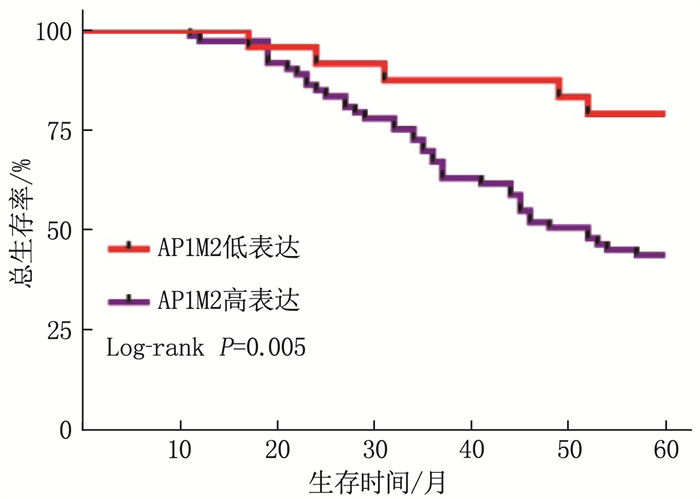
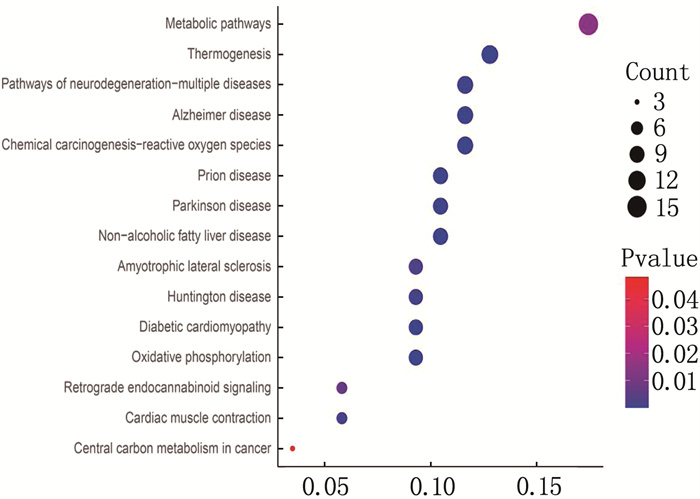
结果免疫组化分析发现, AP1M2在结肠癌组织中的表达高于癌旁组织; 在97对标本中,结肠癌组织AP1M2高表达73例,癌旁组织AP1M2高表达43例,差异有统计学意义(P=0.004)。酶联免疫吸附测定法检测发现,结肠癌患者癌组织血清中AP1M2表达高于癌旁组织,差异有统计学意义(P=0.02)。肿瘤直径>5 cm、N1~N2分期(有淋巴结转移)、T分期的T3~T4期、术后5年复发患者AP1M2高表达比率分别高于肿瘤直径≤5 cm、N0分期(无淋巴结转移)、T分期的T1~T2期、术后5年无复发患者,差异有统计学意义(P < 0.05)。术后随访5年, 24例AP1M2低表达患者术后5年死亡5例,而73例AP1M2高表达患者术后5年死亡24例,差异有统计学意义(P=0.005)。Cox分析显示, AP1M2是结肠癌患者术后死亡的危险因素。生物学分析显示, AP1M2与肿瘤代谢及黏附相关。
结论AP1M2表达水平在结肠癌组织中及血清中上调,这与肿瘤临床分期、淋巴结转移情况相关,与结肠癌患者预后也相关。AP1M2可作为结肠癌预后的潜在预测指标。
Abstract:ObjectiveTo explore the relations of expression of adaptor-related protein complex 1 subunit Mu2 (AP1M2) with clinical pathological features in patients with colorectal carcinoma.
MethodsThe cancer tissues and adjacent tissues of 97 patients with radical surgery for malignant tumor of colon from January to December 2017 in the Lianyungang City Second People′s Hospital were collected. Clinical pathological characteristic materials of patients were collected, the immunohistochemistry was used to detect the expression level of AP1M2 protein in colorectal carcinoma tissues and adjacent tissues, and the enzyme-linked immunosorbent assay was used to detect the expression of AP1M2 in serum. The relationship between the expression of AP1M2 protein and the clinical pathological characteristics in patients with malignant tumor of colon was analyzed; the Kaplan-Meier method was used to analyze the influencing factors of postoperative 5-year survival in patients with colorectal carcinoma; the bioinformatics was used to analyze the proteins and related pathways involved in AP1M2.
ResultsImmunohistochemical analysis showed that the expression of AP1M2 in colon cancer tissues was higher than that in adjacent tissues; among 97 pairs of specimens, there were 73 cases with high expression of AP1M2 in colon cancer tissues and 43 cases with high expression of AP1M2 in adjacent tissues, and there was a significant difference between two kinds of tissues (P=0.004). Enzyme linked immunosorbent assay showed that the expression of serum AP1M2 in cancer tissues of patients with colorectal carcinoma was significantly higher than that in the adjacent tissues (P=0.02). The ratio of high expression of AP1M2 in patients with tumor diameter>5 cm, N staging of N1 to N2 (with lymph node metastasis), T staging of T3 to T4 and postoperative 5-year recurrence was respectively significantly higher than that in patients with tumor diameter≤5 cm, N0 staging (without lymph node metastasis), T1 to T2 of T staging and no postoperative 5-year recurrence (P < 0.05). After 5 years of postoperative follow-up, 5 cases died at 5 years after surgery in 24 patients with low expression of AP1M2, while 24 cases died at 5 years after surgery in 73 patients with high expression of AP1M2, and there was a significant difference (P=0.005). Cox analysis showed that AP1M2 was a risk factor for postoperative mortality of colorectal carcinoma. Biological analysis showed that AP1M2 was associated with tumor metabolism and adhesion.
ConclusionThe expression level of AP1M2 is upregulated in colon cancer tissues and serum, which is related to tumor clinical staging, condition of lymph node metastasis, and prognosis of patients with colorectal carcinoma. AP1M2 can be used as a potential prognostic indicator for colorectal carcinoma.
-
-
表 1 AP1M2在97例结肠癌中的表达情况
组织类型 n AP1M2表达 高表达 低表达 结肠癌组织 97 73* 24* 癌旁组织 97 43 54 与癌旁组织比较, *P < 0.05。 表 2 AP1M2表达与结肠癌临床病理特征的关系
临床病理特征 分类 n AP1M2高表达组 AP1M2低表达组 P 年龄 < 60岁 33 21 12 0.081 ≥60岁 64 52 12 性别 男 59 46 13 0.476 女 38 27 11 肿瘤直径 ≤5 cm 51 33 18 0.017 >5 cm 46 40 6 分化程度 高分化 42 28 14 0.388 中分化 22 16 6 低分化 33 19 4 T分期 T1~T2期 40 23 17 0.001 T3~T4期 57 50 7 N分期 N0期 77 54 23 0.021 N1~N2期 20 19 1 M分期 M0期 84 63 23 0.286 M1期 13 12 1 神经脉管侵犯 有 28 25 3 0.067 无 69 48 21 癌胚抗原/(ng/mL) 97 6.27±10.34 25.98±118.06 0.417 术后5年复发 有 18 17 1 0.036 无 79 56 23 表 3 结肠癌术后5年生存影响因素的多因素分析
因素 HR 95%CI P 下限 上限 T分期 7.492 1.356 41.388 0.021 N分期 0.537 0.214 1.346 0.185 M分期 2.358 0.959 5.798 0.062 分化程度 0.877 0.567 1.355 0.554 神经脉管侵犯 0.206 0.073 0.579 0.003 癌胚抗原 1.002 0.993 1.010 0.686 AP1M2水平 0.243 0.076 0.778 0.017 -
[1] WEITZ J, KOCH M, DEBUS J, et al. Colorectal cancer[J]. Lancet, 2005, 365(9454): 153-165. doi: 10.1016/S0140-6736(05)17706-X
[2] CHEN K, COLLINS G, WANG H, et al. Pathological features and prognostication in colorectal cancer[J]. Curr Oncol, 2021, 28(6): 5356-5383. doi: 10.3390/curroncol28060447
[3] FÖLSCH H, OHNO H, BONIFACINO J S, et al. A novel clathrin adaptor complex mediates basolateral targeting in polarized epithelial cells[J]. Cell, 1999, 99(2): 189-198. doi: 10.1016/S0092-8674(00)81650-5
[4] YI Y X, ZHANG Q F, SHEN Y F, et al. System analysis of adaptor-related protein complex 1 subunit mu 2 (AP1M2) on malignant tumors: a pan-cancer analysis[J]. J Oncol, 2022, 2022: 7945077.
[5] LIU S, CAI Y, CHANGYONG E, et al. Screening and validation of independent predictors of poor survival in pancreatic cancer[J]. Pathol Oncol Res, 2021, 27: 1609868. doi: 10.3389/pore.2021.1609868
[6] ARNOLD M, SIERRA M S, LAVERSANNE M, et al. Global patterns and trends in colorectal cancer incidence and mortality[J]. Gut, 2017, 66(4): 683-691. doi: 10.1136/gutjnl-2015-310912
[7] YANG H J, WU J, ZHANG J J, et al. Integrated bioinformatics analysis of key genes involved in progress of colon cancer[J]. Mol Genet Genomic Med, 2019, 7(4): e00588. doi: 10.1002/mgg3.588
[8] WEN J, MIN X J, SHEN M Q, et al. ACLY facilitates colon cancer cell metastasis by CTNNB1[J]. J Exp Clin Cancer Res, 2019, 38(1): 401. doi: 10.1186/s13046-019-1391-9
[9] WANG D W, YANG Z S, XU J, et al. Identification of prognostic genes for colon cancer through gene Co-expression network analysis[J]. Curr Med Sci, 2021, 41(5): 1012-1022. doi: 10.1007/s11596-021-2386-2
[10] ]VILLALOBOS C, HERNÁNDEZ-MORALES M, GUTIÉRREZ L G, et al. TRPC1 and ORAI1 channels in colon cancer[J]. Cell Calcium, 2019, 81: 59-66. doi: 10.1016/j.ceca.2019.06.003
[11] ZHANG Z Y, JI M L, LI J, et al. Molecular classification based on prognostic and cell cycle-associated genes in patients with colon cancer[J]. Front Oncol, 2021, 11: 636591. doi: 10.3389/fonc.2021.636591
[12] KARPISHEH V, NIKKHOO A, HOJJAT-FARSANGI M, et al. Prostaglandin E2 as a potent therapeutic target for treatment of colon cancer[J]. Prostaglandins Other Lipid Mediat, 2019, 144: 106338. doi: 10.1016/j.prostaglandins.2019.106338
[13] LIU Z Y, YANG L, ZHONG C B, et al. EZH2 regulates H2B phosphorylation and elevates colon cancer cell autophagy[J]. J Cell Physiol, 2020, 235(2): 1494-1503. doi: 10.1002/jcp.29069
[14] AVRUCH J, ZHOU D W, BARDEESY N. YAP oncogene overexpression supercharges colon cancer proliferation[J]. Cell Cycle, 2012, 11(6): 1090-1096. doi: 10.4161/cc.11.6.19453
[15] RASOOL M, NATESAN PUSHPARAJ P, KARIM S. Overexpression of CXCL8 gene in Saudi colon cancer patients[J]. Saudi J Biol Sci, 2021, 28(11): 6045-6049. doi: 10.1016/j.sjbs.2021.09.031
[16] WU H W, FAN L J, LIU H P, et al. Identification of key genes and prognostic analysis between chromophobe renal cell carcinoma and renal oncocytoma by bioinformatic analysis[J]. Biomed Res Int, 2020, 2020: 4030915.
[17] COLAS E, PEREZ C, CABRERA S, et al. Molecular markers of endometrial carcinoma detected in uterine aspirates[J]. Int J Cancer, 2011, 129(10): 2435-2444. doi: 10.1002/ijc.25901




 下载:
下载:



 苏公网安备 32100302010246号
苏公网安备 32100302010246号