Regulatory role and mechanism of mitochondrial ribosomal protein S35 in proliferation, invasion, and migration of colon cancer cells
-
摘要:目的
探讨线粒体核糖体蛋白S35(MRPS35)对结肠癌细胞增殖、侵袭和迁移的调控作用及机制。
方法收集120例结肠癌根治术患者的结肠癌组织及癌旁正常组织标本, 培养人结肠癌细胞系(HCT116、SW480、SW620)和人正常结肠上皮细胞系(NCM460)。通过生物信息学分析、实时荧光定量聚合酶链反应、蛋白质印迹法(Western blot)、免疫组织化学(IHC)分析、细胞功能实验(平板克隆形成实验、划痕实验、Transwell细胞迁移实验、CCK-8细胞活力实验)等方法评估MRPS35在结肠癌中的表达及调控机制。
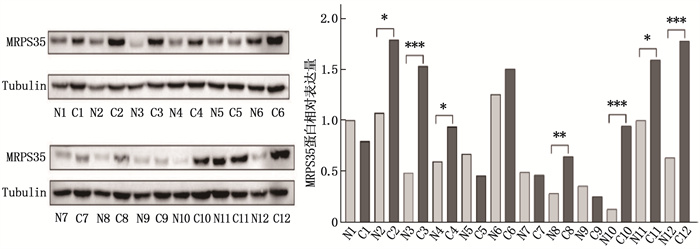
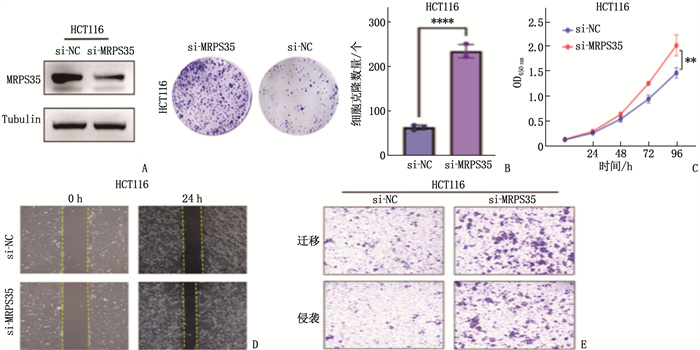
结果生物信息学分析结果显示, MRPS35基因在结直肠癌组织中的表达水平高于癌旁正常组织,差异有统计学意义(P < 0.05)。人结肠癌细胞系(HCT116、SW480、SW620)中MRPS35 mRNA和MRPS35蛋白相对表达量均高于NCM460细胞,差异有统计学意义(P < 0.05); 结肠癌组织中的MRPS35蛋白相对表达量高于癌旁正常组织,差异有统计学意义(P < 0.05)。MRPS35表达水平与肿瘤直径、肿瘤分化程度、T分期显著相关(P=0.002、0.021、0.036)。MRPS35高表达患者的总体生存率高于MRPS35低表达患者,差异有统计学意义(Log-rank P=0.015)。敲低MRPS35后,结肠癌细胞克隆、增殖、侵袭、迁移能力均显著增强。敲低MRPS35后, Wnt1、β-Catenin及其下游靶标蛋白的表达显著增加。
结论MRPS35在结肠癌组织和结肠癌细胞中均显著高表达,其可能通过调控Wnt/β-Catenin信号通路抑制结肠癌的发生与发展,有望成为结肠癌的新型生物标志物和潜在治疗靶点。
-
关键词:
- 线粒体核糖体蛋白S35 /
- Wnt/β-Catenin信号通路 /
- 结肠癌细胞 /
- 增殖 /
- 侵袭 /
- 迁移
Abstract:ObjectiveTo investigate the regulatory role and mechanism of mitochondrial ribosomal protein S35 (MRPS35) in the proliferation, invasion, and migration of colon cancer cells.
MethodsA total of 120 colon cancer tissues and adjacent normal tissues from patients undergoing radical resection for colon cancer were collected. Human colon cancer cell lines (HCT116, SW480, SW620) and a human normal colon epithelial cell line (NCM460) were cultured. Bioinformatics analysis, real-time quantitative polymerase chain reaction, Western blot, immunohistochemical (IHC) analysis, and cellular functional experiments (plate clone formation assay, scratch test, Transwell migration assay, CCK-8 cell viability assay) were conducted to evaluate the expression and regulatory mechanism of MRPS35 in colon cancer.
ResultsBioinformatics analysis showed that the expression level of the MRPS35 gene was higher in colorectal cancer tissues than in adjacent normal tissues (P < 0.05). The relative expression levels of MRPS35 mRNA and MRPS35 protein were higher in human colon cancer cell lines (HCT116, SW480, SW620) than in NCM460 cells (P < 0.05). The relative expression level of MRPS35 protein was higher in colon cancer tissues than that in adjacent normal tissues (P < 0.05). The expression level of MRPS35 was significantly correlated with tumor diameter, tumor differentiation, and T stage (P=0.002, 0.021, 0.036). Patients with high MRPS35 expression had a higher overall survival rate than those with low MRPS35 expression (Log-rank P=0.015). After knockdown of MRPS35, the abilities of colon cancer cell cloning, proliferation, invasion, and migration were significantly enhanced. Furthermore, the expression of Wnt1, β-Catenin, and their downstream target proteins increased significantly after MRPS35 knockdown.
ConclusionMRPS35 is significantly overexpressed in both colon cancer tissues and colon cancer cells, and it may inhibit the occurrence and development of colon cancer by regulating the Wnt/β-Catenin signaling pathway. Therefore, MRPS35 has the potential to become a novel biomarker and therapeutic target for colon cancer.
-
-
图 2 基于TCGA数据库分析MRPS35基因在泛癌组织中的表达情况
BLCA: 膀胱癌; BRCA: 乳腺癌易感基因; CESC: 宫颈鳞状细胞癌; CHOL: 胆管癌; COAD: 结肠腺癌; ESCA: 食管癌; GBM: 胶质母细胞瘤; HNSC: 头颈部鳞状细胞癌; KICH: 肾嫌色细胞癌; KIRC: 肾透明细胞癌; KIRP: 肾乳头状细胞癌; LIHC: 肝细胞肝癌; LUAD: 肺腺癌; LUSC: 肺鳞状细胞癌; PAAD: 胰腺腺癌; PRAD: 前列腺腺癌; PCPG: 嗜铬细胞瘤和副神经节瘤; READ: 直肠腺癌; SARC: 肉瘤; SKCM: 皮肤黑色素瘤; THCA: 甲状腺癌; THYM: 胸腺瘤; STAD: 胃腺癌; UCEC; 子宫内膜癌。蓝色表示癌旁正常组织,红色表示癌组织。
表 1 PCR引物序列
基因 正向引物(5′→3′) 反向引物(5′→3′) MRPS35 GCCCAGAACCCATTTCCTTGAC GGACCTGTTTCTAACCAGAGTTGAC β-actin ATCGTGCGTGACATTAAGGAGAAG AGGAAGGAAGGCTGGAAGAGTG 表 2 不同MRPS35表达水平结肠癌患者的临床特征比较[n(%)]
临床特征 分类 n MRPS35高表达组(n=77) MRPS35低表达组(n=43) P 年龄 < 65岁 37 24(31.2) 13(30.2) 0.975 ≥65岁 83 53(68.8) 30(69.8) 性别 男 99 60(77.9) 39(90.7) 0.204 女 21 17(22.1) 4(9.3) 肿瘤直径 < 5 cm 77 50(64.9) 27(62.8) 0.002 ≥5 cm 43 27(35.1) 16(37.2) 肿瘤分化程度 中高分化 67 45(58.4) 22(51.2) 0.021 低分化 53 32(41.6) 21(48.8) T分期 T1~T2期 68 45(58.4) 23(53.5) 0.036 T3~T4期 52 32(41.6) 20(46.5) N分期 N0期 67 43(55.8) 24(55.8) 0.624 N1~N2期 53 34(44.2) 19(44.2) M分期 M0期 111 70(90.9) 41(95.3) 0.447 M1期 9 7(9.1) 2(4.7) -
[1] SUNG H, FERLAY J, SIEGEL R L, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660
[2] FIDLER M M, SOERJOMATARAM I, BRAY F. A global view on cancer incidence and national levels of the human development index[J]. Int J Cancer, 2016, 139(11): 2436-2446. doi: 10.1002/ijc.30382
[3] KOPPE M J, BOERMAN O C, OYEN W J, et al. Peritoneal carcinomatosis of colorectal origin: incidence and current treatment strategies[J]. Ann Surg, 2006, 243(2): 212-222. doi: 10.1097/01.sla.0000197702.46394.16
[4] LIN X Y, GUO L J, LIN X, et al. Expression and prognosis analysis of mitochondrial ribosomal protein family in breast cancer[J]. Sci Rep, 2022, 12: 10658. doi: 10.1038/s41598-022-14724-7
[5] LI X, WANG M, LI S, et al. HIF-1-induced mitochondrial ribosome protein L52: a mechanism for breast cancer cellular adaptation and metastatic initiation in response to hypoxia[J]. Theranostics, 2021, 11(15): 7337-7359. doi: 10.7150/thno.57804
[6] CHARAFE-JAUFFRET E, GINESTIER C, IOVINO F, et al. Breast cancer cell lines contain functional cancer stem cells with metastatic capacity and a distinct molecular signature[J]. Cancer Res, 2009, 69(4): 1302-1313. doi: 10.1158/0008-5472.CAN-08-2741
[7] SIEGEL R L, MILLER K D, GODING SAUER A, et al. Colorectal cancer statistics, 2020[J]. CA A Cancer J Clinicians, 2020, 70(3): 145-164. doi: 10.3322/caac.21601
[8] BAI Z, XIA X, LU J. microRNA-639 is down-regulated in hepatocellular carcinoma tumor tissue and inhibits proliferation and migration of human hepatocellular carcinoma cells through the KAT7/wnt/β-catenin pathway[J]. Med Sci Monit, 2020, 26: e919241.
[9] YUAN Y, JU Y S, KIM Y, et al. Comprehensive molecular characterization of mitochondrial genomes in human cancers[J]. Nat Genet, 2020, 52: 342-352. doi: 10.1038/s41588-019-0557-x
[10] 3LIU L, LUO C, LUO Y, et al. MRPL33 and its splicing regulator hnRNPK are required for mitochondria function and implicated in tumor progression[J]. Oncogene, 2018, 37(1): 86-94. doi: 10.1038/onc.2017.314
[11] LEE Y K, LIM J J, JEOUN U W, et al. Lactate-mediated mitoribosomal defects impair mitochondrial oxidative phosphorylation and promote hepatoma cell invasiveness[J]. J Biol Chem, 2017, 292(49): 20208-20217. doi: 10.1074/jbc.M117.809012
[12] TANG B, ZHU J, ZHAO Z, et al. Diagnosis and prognosis models for hepatocellular carcinoma patient's management based on tumor mutation burden[J]. J Adv Res, 2021, 33: 153-165. doi: 10.1016/j.jare.2021.01.018
[13] 黄灿坡, 王琳, 林建泉, 等. circMRPS35和KAT7在胃癌组织中的表达及预后评估价值[J]. 国际检验医学杂志, 2022, 43(23): 2846-2851. [14] PU M, WANG J L, HUANG Q K, et al. High MRPS23 expression contributes to hepatocellular carcinoma proliferation and indicates poor survival outcomes[J]. Tumour Biol, 2017, 39(7): 101042831770912.
[15] WANG X, PERALTA S, MORAES C T. Mitochondrial alterations during carcinogenesis: a review of metabolic transformation and targets for anticancer treatments[J]. Adv Cancer Res, 2013, 119: 127-160.
[16] PIAO L, LI Y, KIM S J, et al. Association of LETM1 and MRPL36 contributes to the regulation of mitochondrial ATP production and necrotic cell death[J]. Cancer Res, 2009, 69(8): 3397-3404. doi: 10.1158/0008-5472.CAN-08-3235
[17] CHEN Y C, CHANG M Y, SHIAU A L, et al. Mitochondrial ribosomal protein S36 delays cell cycle progression in association with p53 modification and p21(WAF1/CIP1) expression[J]. J Cell Biochem, 2007, 100(4): 981-990. doi: 10.1002/jcb.21079
[18] YOO Y A, KIM M J, PARK J K, et al. Mitochondrial ribosomal protein L41 suppresses cell growth in association with p53 and p27Kip1[J]. Mol Cell Biol, 2005, 25(15): 6603-6616. doi: 10.1128/MCB.25.15.6603-6616.2005
[19] TANIUE K, HAYASHI T, KAMOSHIDA Y, et al. UHRF1-KAT7-mediated regulation of TUSC3 expression via histone methylation/acetylation is critical for the proliferation of colon cancer cells[J]. Oncogene, 2020, 39: 1018-1030. doi: 10.1038/s41388-019-1032-y
[20] CLEVERS H. Wnt/beta-catenin signaling in development and disease[J]. Cell, 2006, 127(3): 469-480. doi: 10.1016/j.cell.2006.10.018
[21] KRAUSOVA M, KORINEK V. Wnt signaling in adult intestinal stem cells and cancer[J]. Cell Signal, 2014, 26(3): 570-579.





 下载:
下载:







 苏公网安备 32100302010246号
苏公网安备 32100302010246号
